New insights on molecular mechanism of hydrocephalus could lead to first ever non-surgical treatment
- johnshohfi
- Mar 8, 2023
- 3 min read
Updated: Aug 23, 2023
Original Article Here by McKenzie Ridings
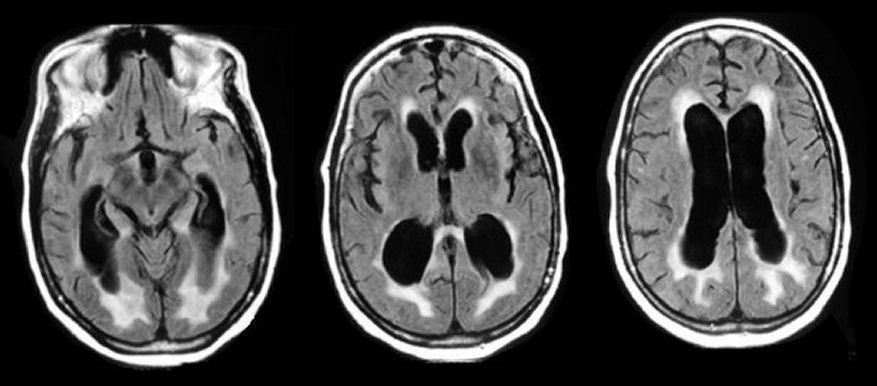
Mass General researchers have discovered a novel molecular mechanism responsible for the most common forms of acquired hydrocephalus, potentially opening the door to the first-ever nonsurgical treatment for a life-threatening disease that affects about a million Americans. As reported in the journal Cell, the team uncovered in animal models the pathway through which infection or bleeding in the brain triggers a massive neuroinflammatory response that results in increased production of cerebrospinal fluid (CSF) by tissue known as the choroid plexus, leading to swelling of the brain ventricles.
“Finding a nonsurgical treatment for hydrocephalus, given the fact neurosurgery is fraught with tremendous morbidity and complications, has been the holy grail for our field,” says Kristopher Kahle, MD, PhD, a pediatric neurosurgeon at MGH and senior author of the study. “We’ve identified through a genome-wide analytical approach the mechanism that underlies the swelling of the ventricles which occurs after a brain bleed or brain infection in acquired hydrocephalus. We’re hopeful these findings will pave the way for approval of an anti-inflammatory drug to treat hydrocephalus, which could be a game-changer for populations in the U.S. and around the world that don’t have access to surgery.”
Acquired hydrocephalus occurs in about one of every 500 births globally. It is the most common cause of brain surgery in children, though it can affect people at any age. In underdeveloped parts of the world where bacterial infection is the most prevalent form of the disease, hydrocephalus is often deadly for children due to the lack of surgical intervention. Indeed, the only known treatment for acquired hydrocephalus is brain surgery, which involves implantation of a catheter-like shunt to drain fluid from the brain. But about half of all shunts in pediatric patients fail within two years of placement, according to the Hydrocephalus Association, requiring repeat neurosurgical operations and a lifetime of brain surgeries.
By deciphering the unique cellular and molecular biology that occurs within the brain after infection or severe hemorrhage, the MGH-led research team has taken a major step toward nonsurgical, pharmacologic treatment for humans. Pivotal to the process is the choroid plexus, the brain structure that routinely pumps cerebrospinal fluid into the four ventricles of the brain to keep the organ buoyant and injury-free within the skull. An infection or brain bleed, however, can create a dangerous neuroinflammatory response where the choroid plexus floods the ventricles with cerebral spinal fluid and immune cells from the periphery of the brain – a so-called “cytokine storm,” or immune system overreaction, so often seen in COVID-19 infections--swelling the brain ventricles.
“Scientists in the past thought that entirely different mechanisms were involved in hydrocephalus from infection and from hemorrhage in the brain,” explains co-author Bob Carter, MD, PhD, chair of the Department of Neurosurgery at MGH. “Dr. Kahle’s lab found that the same pathway was involved in both types and that it can be targeted with immunomodulators like rapamycin, a drug that’s been approved by the U.S. Food and Drug Administration for transplant patients who need to suppress their immune system to prevent organ rejection.”
MGH researchers are continuing to explore how rapamycin and other repurposed drugs which quell the inflammation seen in acquired hydrocephalus could be turned into an effective drug treatment for patients. “What has me most excited is that this noninvasive therapy could provide a way to help young patients who don’t have access to neurosurgeons or shunts,” emphasizes Kahle. “No longer would a diagnosis of hydrocephalus be fatal for these children.”
Kahle is director of Pediatric Neurosurgery at MGH, and director of the Harvard Center for Hydrocephalus and Neurodevelopmental Disorders. Carter is chief of Neurosurgery Service at MGH and professor of Surgery at Harvard Medical School.
The study was funded by the National Institutes of Health and the Hydrocephalus Association.



Comments